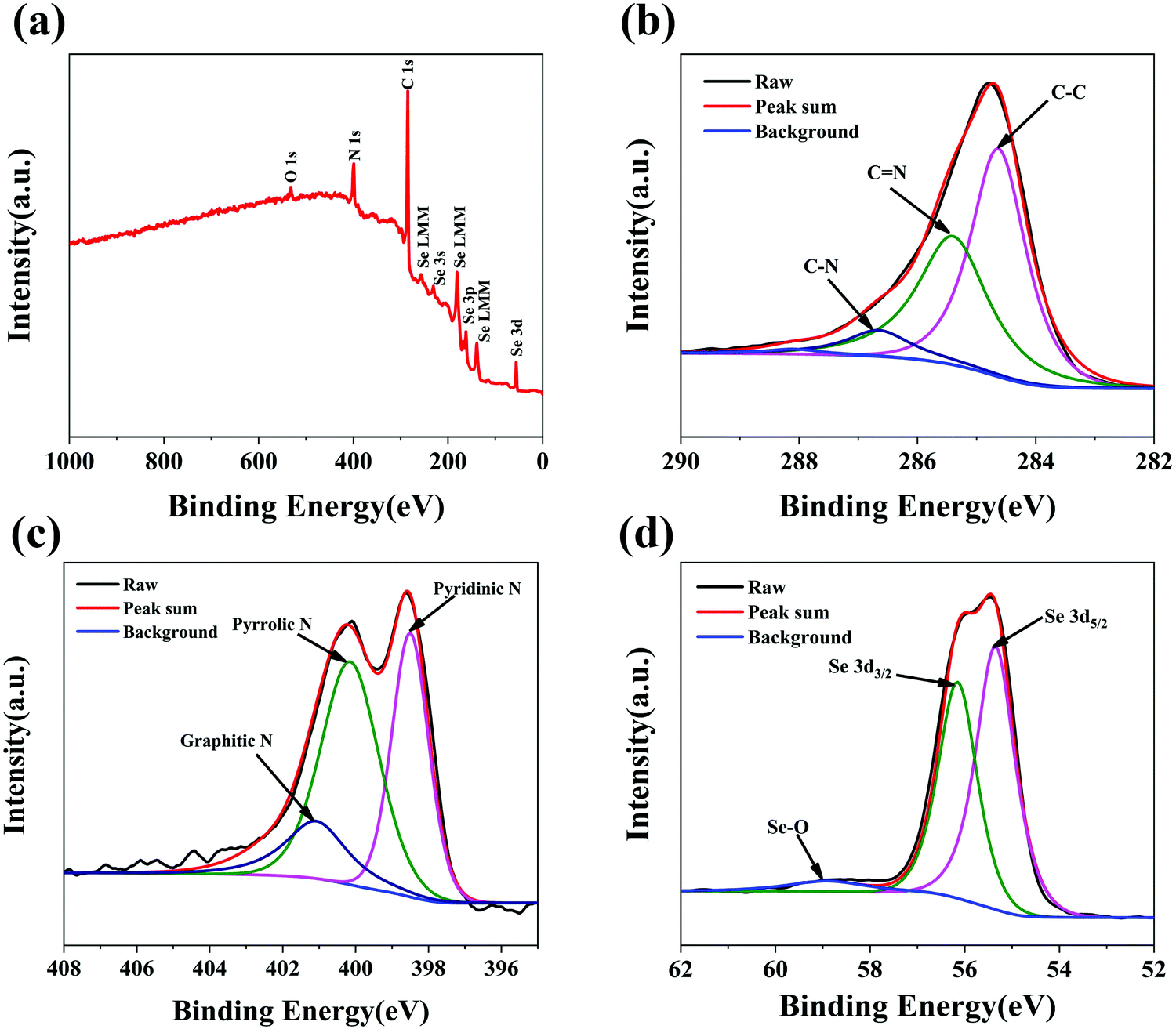
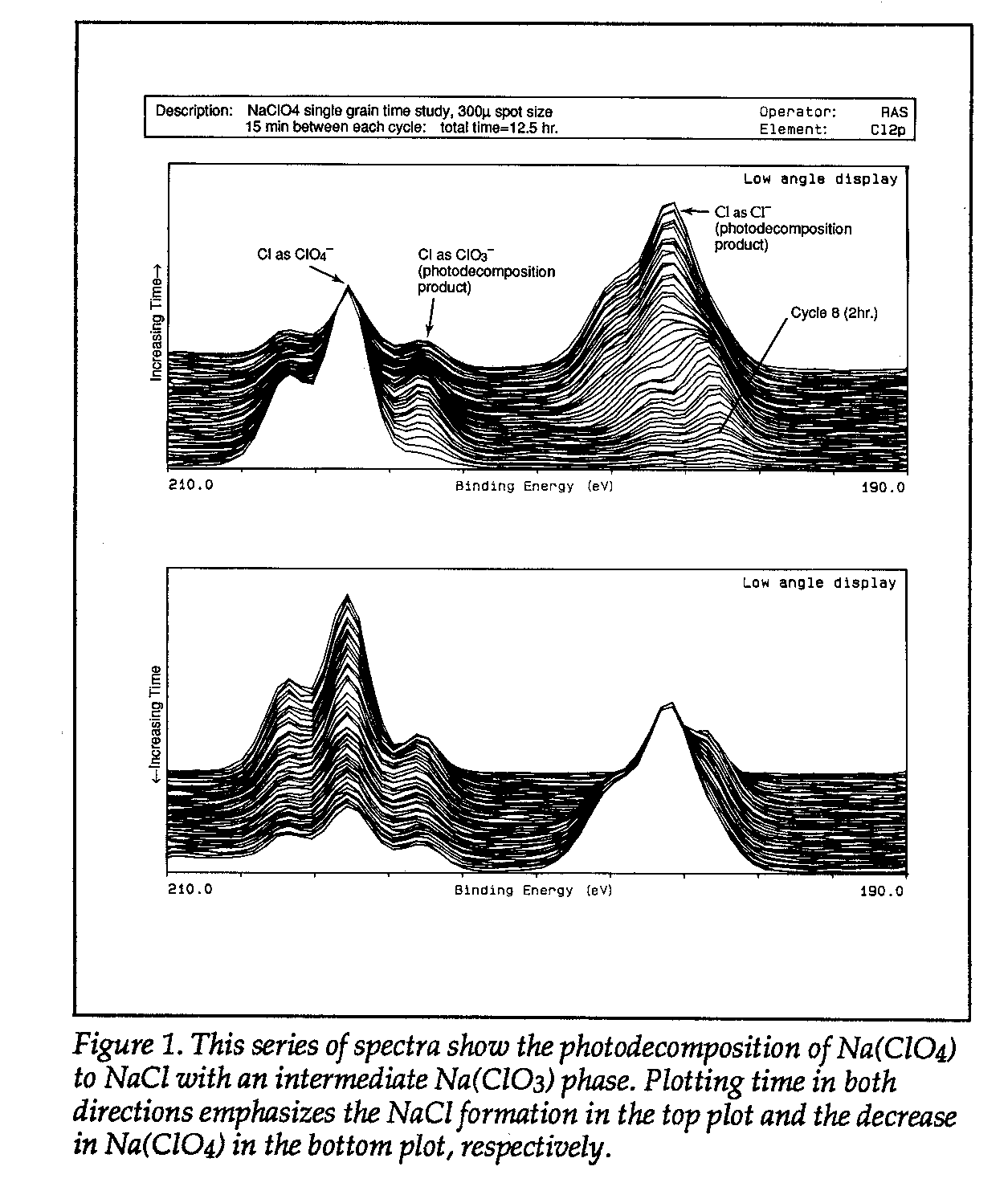
Naclo4 xps peak sodium ion
"Chlorine Oxides and Chlorine Oxygen Acids". One of the first attempt of a RT sodium solid-state batteries employing NASICON electrolyte was reported by Noguchi et al., fabricating an all-solid-state sodium-ion symmetrical battery via combined screen printing and hot pressing using Na 3 Zr 2 Si 2 PO 12 (NASICON) as solid electrolyte and Na 3 V 2 (PO 4) 3 (NVP) as active electrode materials.
Explore the world of surface science, nanotechnologies, materials science and scientific research instrumentations. Bennett, Peter Wintzer, Saeed Akbar Sheikh, Patrizio Gallone (2002). Scienta Omicron is a leading innovator in surface science and nanotechnology that provides top capabilities in electron spectroscopy, scanning probe microscopy and thin film deposition, all in ultra-high vacuum (UHV). "De l'eau liquide répérée sur les pentes martiennes". "Perchlorates on Mars enhance the bacteriocidal effects of UV light". ^ Wadsworth, Jennifer Cockell, Charles S.ClO 3 − + H 2O → ClO 4 − + 2H + + 2 e − (acidic medium) ClO 3 − + 2 OH − → ClO 4 − + H 2O + 2 e − (alkaline medium) Safety Sodium perchlorate is produced by anodic oxidation of sodium chlorate at an inert electrode, such as platinum.
Sodium perchlorate can be used to block iodine uptake before administration of iodinated contrast agents in patients with subclinical hyperthyroidism (suppressed TSH). It is used in standard DNA extraction and hybridization reactions in molecular biology. Solutions of NaClO 4 are often used as an unreactive electrolyte. These reference pages contain tips and techniques that are designed to help both the novice and advanced XPS user. Perchloric acid is made by treating NaClO 4 with HCl.Īmmonium perchlorate and potassium perchlorate, of interest in rocketry and pyrotechnics, are prepared by double decomposition from a solution of sodium perchlorate and ammonium chloride or potassium chloride, respectively. X-ray photoelectron spectroscopy (XPS or ESCA) curve fitting procedures, reference materials and useful notes are listed here to provide a starting point for the consistent interpretation of XPS spectra. Sodium perchlorate is the precursor to many other perchlorate salts, often taking advantage of their low solubility relative to NaClO 4 (209 g/100 mL at 25 ☌). It crystallizes in the rhombic crystal system. it is favorable for it to decompose into sodium chloride and dioxygen. Its heat of formation is −382.75 kJ/mol, i.e.
